Understanding Multiple Sclerosis (MS)
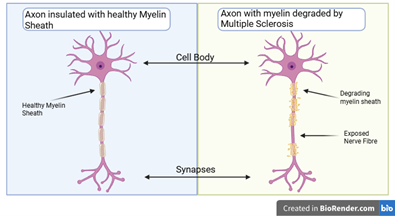
Multiple Sclerosis (MS) is an autoimmune disorder that affects about 2.8 million people worldwide 1. The diseases main mechanism of action is to trigger immune cells such as macrophages and T-cells to break down the myelin sheath 2. The myelin sheath protects neurons, allowing neurological signals to travel quickly and safely through the central nervous system (CNS). The degradation of the myelin sheath leads to the development of lesions throughout the CNS. These lesions can cause surrounding nerve cells to die off at accelerated rates, interrupting signal transduction. MS symptoms depend on the location and severity of lesions. For example, lesions on the brain and spinal cord can impair motor function and autonomy 3.
figure 1: Visualisation of the degradation of the myelin sheath caused by MS. Made with Biorender4
Symptoms of MS can vary greatly and progress over time. They generally include2:
-
Fatigue
-
Numbness and tingling
-
Visual impairment
-
Bladder and bowel dysfunction
-
Cognitive impairment
Possible Causes and Risk Factors
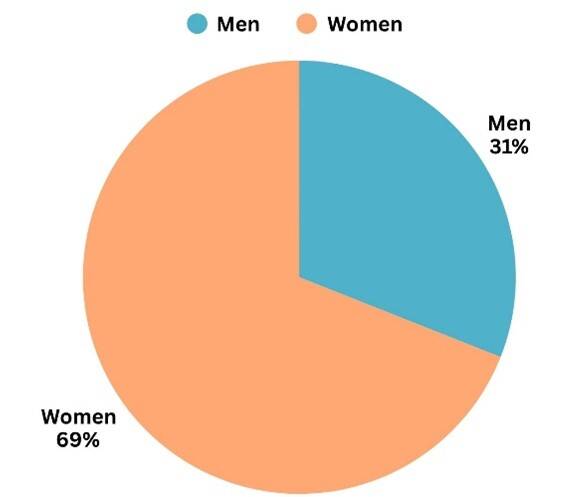
The causes behind MS are unclear 5. However, some genetic and environmental factors, including living far north of the equator, smoking, obesity, and low vitamin D levels show a correlation in MS 6. The largest risk factor for MS is gender as MS is over twice as prevalent in women than in men. The MS International Foundation reports that over 69% of MS cases are in women1, 7.
figure 2: Ratio of Men to Women affected by MS. Made with Canva22
Case Study: Postpartum Relapse in a Woman with MS
The aim of this blog is to look into a case study of a woman with MS who had just had a baby. She took teriflunomide(Aubagio) daily before the pregnancy and stopped taking it at about week 15. While she intended to breastfeed the baby, she had an MS relapse about 1 month postpartum and was advised to go back on teriflunomide. She was informed she should not be breast feeding while on teriflunomide.
Disease-Modifying Drugs (DMDs) and Pregnancy
Women with MS commonly stop taking disease-modifying drugs (DMD) during pregnancy due to the lack of information available about their safety 8. In the case of teriflunomide, while no human studies are available, teriflunomide labels often cite that it can excrete into breast milk in animal studies 9, 10, 11. Moreover, the side effects of teriflunomide, such as diarrhoea, hair thinning, influenza, and nausea 12, are generally manageable for adults, but are too severe for a newborn or developing foetus to risk passing the drug onto them.
Postpartum Relapse Risk
Findings in a meta-analysis by Charlotte Schubert13 suggest that the chances of a relapse are significantly higher in the first 3 months postpartum compared to the pre-pregnancy period. Furthermore, patients who took DMD for MS before pregnancy were more than twice as likely to experience a relapse in the first 3 months postpartum. While patients who exclusively breastfeed showed a reduced risk of relapse, more data is needed to confirm this effect.
One of the key physiological changes postpartum is the reactivation of the immune system. During pregnancy, pregnancy hormones such as oxytocin and progesterone cause a shift in the immune system. 14. This shift decreases the expression of cell mediated activity, which is a non-specific process for neutralising abnormal cells, and an increase in humoral immunity, where specific antigens found on viruses and antibodies are targeted 15. These changes protect the foetus from being recognised as a foreign body by the innate immune system. This process is believed to also downregulate the autoimmune activity of MS during the pregnancy, which could be why there is relief in MS symptoms without the need for DMDs 14.
The Role of Estriol and Hormonal Changes
Estriol is a placenta-made oestrogen hormone that expressed progressively more throughout a pregnancy. As one of the hormones involved in the shift to humoral immunity, estriol promotes anti-inflammatory activity, protecting the foetus and reducing MS activity 16. After pregnancy, estriol production shows a drastic decline without the placenta. The decrease in estriol is a contributing factor to the immune system reversing to more normal activity. In the case of MS patients, this can lead to an increase in autoimmune myelin breakdown and unfortunately, increases the risk of relapse by MS significantly 14. This is a possible reason as to why the woman in this case study had a relapse so early in the postpartum period. She likely started taking the DMD again to suppress the cell-mediated immune response.
Preventive Measures and Safer Alternatives
MS patients who are trying for conception should coordinate with their doctor. Several precautions should have been taken to prevent the outcome of this case study and reduce the risk of potential harm to the foetus. There are alternative DMDs for MS that are considered safe to take during pregnancy and the breast-feeding. Glatiramer acetate is a novel treatment for the autoimmune effects of MS 17 that has shown no adverse effects on breast-fed infants 18. Many DMD’s for MS have long washout periods. Teriflunomide can take up to 2 years to be completely eliminated from blood plasma 19. Because she was taking teriflunomide so far into the pregnancy, she should have been prescribed cholestyramine or activated charcoal, which stimulate a rapid washout of teriflunomide in the blood to reduce the risk of passing any teratogenic or hepatotoxic effects onto the foetus 20. She also could have benefited from being recommended an obstetrician to screen for any complications that may come up 21.
Conclusion
Multiple sclerosis poses significant challenges for women who are considering pregnancy, are currently pregnant, or are in the postpartum period. Having to make decisions that could impact their infant or their own life, such as going back on DMD postpartum, stopping their DMD before or during pregnancy, or taking the risk to breastfeed without DMD treatment. While the hormonal changes that occur during pregnancy – such as the immunosuppressive effects of oestrogens like Estriol – can alleviate some of these concerns, it is difficult to deal with the rapid shift back to conditions that increase the risk MS relapse. Fortunately, DMDs such as glatiramer acetate, which have far more favourable data available for their safety during not only pregnancy, but also during breastfeeding. This highlights the need for more research in this area, which could reap huge rewards for women with MS who wish to have children.
Bibliography
- Atlas of MS 2020 - Epidemiology report - MS International Federation. MS International Federation. Published October 26, 2022. https://www.msif.org/resource/atlas-of-ms-2020
- Tafti D, Ehsan M, Xixis KL. Multiple Sclerosis. PubMed. Published 2023. https://www.ncbi.nlm.nih.gov/books/NBK499849
- Cleveland Clinic. Myelin Sheath: What It Is, Purpose & Function. Cleveland Clinic. Published May 9, 2022. https://my.clevelandclinic.org/health/body/22974-myelin-sheath
- Biorender. Published 2025. https://biorender.com
- D’Anca M, Buccellato FR, Gianluca Martino Tartaglia, et al. Why Is Multiple Sclerosis More Frequent in Women? Role of the Immune System and of Oral and Gut Microbiota. Applied Sciences. 2023;13(10):5881-5881. doi:https://doi.org/10.3390/app13105881
- Empowering people affected by MS to live their best lives. National Multiple Sclerosis Society. https://www.nationalmssociety.org/understanding-ms/what-is-ms/who-gets-ms
- Khan G, Hashim MJ. Epidemiology of Multiple Sclerosis: Global, Regional, National and Sub-National-Level Estimates and Future Projections. Journal of Epidemiology and Global Health. 2025;15(1). doi:https://doi.org/10.1007/s44197-025-00353-6
- Disease Modifying Therapies | MS Ireland. www.ms-society.ie. Published February 4, 2019. https://www.ms-society.ie/about-ms/living-ms/treating-and-managing-ms/disease-modifying-therapies
- NDA 202992 -FDA Approved Labeling Text Dated HIGHLIGHTS of PRESCRIBING INFORMATION.; 2012. Accessed October 30, 2025. https://www.accessdata.fda.gov/drugsatfda_docs/label/2012/202992s000lbl.pdf
- Pr S, Teriflunomide. Sandoz Teriflunomide (Teriflunomide Tablets) PRODUCT MONOGRAPH INCLUDING PATIENT MEDICATION INFORMATION.
- Teriflunomide Tablets 14 Mg AUBAGIO. Accessed October 30, 2025. https://www.sanofi.com/assets/countries/india/docs/science-and-innovation/for-healthcare-professionals/product-information/pharmaceuticals/aubagio-pi.pdf
- Aubagio. Accessed October 30, 2025. https://www.ms-society.ie/sites/default/files/2019-06/MS%20Ireland%20Aubagio%20Information%20Sheet%20February%202016.pdf
- Schubert C, Steinberg L, Peper J, et al. Postpartum relapse risk in multiple sclerosis: a systematic review and meta-analysis. Journal of Neurology, Neurosurgery, and Psychiatry. Published online February 20, 2023:jnnp–2022-330533. doi:https://doi.org/10.1136/jnnp-2022-330533
- McCombe P. The Short and Long-Term Effects of Pregnancy on Multiple Sclerosis and Experimental Autoimmune Encephalomyelitis. Journal of Clinical Medicine. 2018;7(12):494. doi:https://doi.org/10.3390/jcm7120494
- Bsteh G, Harald Hegen, Riedl K, et al. Estimating Risk of Multiple Sclerosis Disease Reactivation in Pregnancy and Postpartum: The VIPRiMS Score. Frontiers in Neurology. 2022;12. doi:https://doi.org/10.3389/fneur.2021.766956
- Vermillion MS, Ursin RL, Attreed SE, Klein SL. Estriol Reduces Pulmonary Immune Cell Recruitment and Inflammation to Protect Female Mice From Severe Influenza. Endocrinology. 2018;159(9):3306-3320. doi:https://doi.org/10.1210/en.2018-00486
- Schrempf W, Ziemssen T. Glatiramer acetate: Mechanisms of action in multiple sclerosis. Autoimmunity Reviews. 2007;6(7):469-475. doi:https://doi.org/10.1016/j.autrev.2007.02.003
- Glatiramer. Nih.gov. Published June 15, 2025. Accessed October 30, 2025. https://www.ncbi.nlm.nih.gov/books/NBK501167
- Fuerte-Hortigón A, López Ruiz R, Hiraldo J, et al. Rebound after discontinuation of teriflunomide in patients with multiple sclerosis: 2 case reports. Multiple Sclerosis and Related Disorders. 2020;41:102017. doi:https://doi.org/10.1016/j.msard.2020.102017
- Subei AM, Ontaneda D. Risk Mitigation Strategies for Adverse Reactions Associated with the Disease-Modifying Drugs in Multiple Sclerosis. CNS Drugs. 2015;29(9):759-771. doi:https://doi.org/10.1007/s40263-015-0277-4
- Management of Multiple Sclerosis During Pregnancy. Cleveland Clinic. https://my.clevelandclinic.org/departments/neurological/depts/multiple-sclerosis/ms-approaches/management-of-ms-during-pregnancy
- canva. Free templates | Canva. Canva. Published 2025. https://www.canva.com/templates


Add comment
Comments
Great post! If the estriol is simply returning to pre-pregnancy levels, why do you think that imposes a greater than pre-pregnancy risk of relapse? In my post, I suggested desensitization as a possible mechanism, but I'm curious as to your thoughts on wha the major contributors might be.
Hi Stephen! I Think desensitization is a really novel idea. I think a possible reason for the increased chances of relapse are because generally, the disease-modifying drug (DMD) is not being taken when hormone levels return to normal. The combination of the drop-off in pregnancy hormones and the lack of DMD could mean that she simply wouldn't have the immunoprotective properties asscociated with pregnancy hormones or DMD present in her body. I don't think these ideas contradict eachother too! The increased risk of relapse could be due to a combination of these concepts.