Your baby might have saved your life! At least for about 9 months or 12, then the MS relapses came back again. It´s scary and painful to fear for your ability to take care of your newborn child and oftentimes having to make a tough decision between continuing therapy vs. breastfeeding and ensuring your infant´s health and an important mother-child bond.
But how did your baby save you from Multiple Sclerosis relapses, anyway? Well, the truth is, the baby didn’t, not actively at least. For a successful pregnancy a mother's immune system needs to become tolerant of this recent well-beloved invader. The immune system does not recognize these new foreign cells, at least not inherently, but the placenta takes care of that. The placenta starts signaling to the mother´s immune system using billions of nanosized cargo bubbles, called “exosomes”. These exosomes calm the hyper alert immune system down and let the body know that there is no fatal threat on the door 1 ,3 ,11. That process is supported by a surge in pregnancy hormones like progesterone and estrogen which maintains this tolerant environment and supports the pregnancy 2, 3, 20.
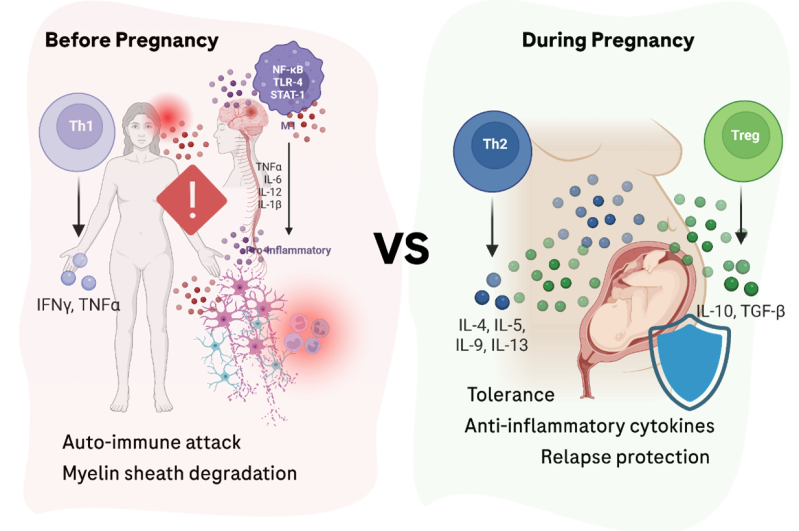
As Multiple sclerosis is assumed to be an autoimmune disease where your own confused immune cells cause inflammation in the nervous system and eat slowly at your neurons 7, 8, it seems like this “relaxed” immune system state and tolerance is a trick to stopping the relentless attacks at the neurons and gives the nerves a chance to take a breath and rebuild again their cover, the myelin sheath, after being torn by your own immune cells.
Figure 1. On the left, the autoimmune intolerant state of multiple sclerosis characterized by an imbalanced Th1 dominant immune system and pro-inflammatory cytokines like IL-6 and on the right, the tolerant pregnancy immune system characterized by a shift towards Th2 immune cells and anti-inflammatory cytokines like IL-10.
How do the exosomes sent by the placenta achieve this tolerant environment in the body?
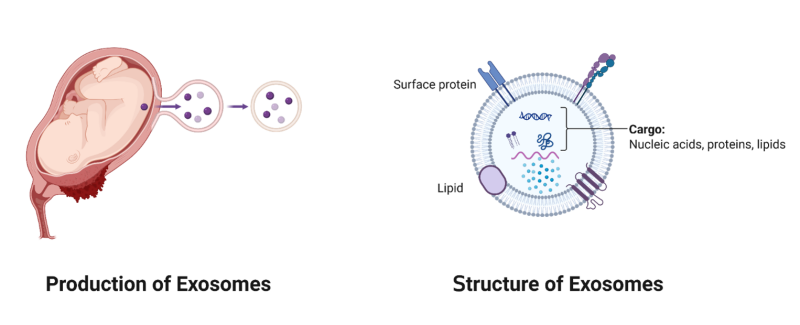
It´s a mixture of tools they carry inside of them and on their own surface 6. On their surface, these exosomes present a protein called PD-L1, Programmed Death-Ligand 1. When the angry immune T-cells come in touch with this protein they receive a “stop attack” message 4, 20.
The placenta doesn't only play defense, however; a baby is on the line after all!
Exosomes themselves could also attack back, they carry other proteins like FasL, Fas Ligand, and TRAIL. When these proteins bind to an activated, aggressive T-cell, they send a "self-destruct" signal, causing the cell to undergo apoptosis aka cell death. This way the population of aggressor cells is selectively controlled and brought down in order for the peaceful ones to outnumber and overpower them 11. It´s not enough to stop the attack, maintaining a regulated system needs recruiting specific T cells called Tregs to keep their own in control 10. This is done using other material carried within the exosomes called microRNAs or miRNAs, supported by the elevated hormones like estrogen. miRNAs can flip switches inside the aggressive T cells to recruit more Tregs and shift from the pro-inflammatory Th1 "attack" mode to an anti-inflammatory Th2 "heal" mode. Tregs find over-active immune cells like the ones we call Th1 and Th17, and suppress them 5. In MS, there's usually an imbalance between the Tregs and Th17s. As the Th17 outnumber the Tregs by folds, it's important that the placenta brings back this balance 3.
Figure 2. On the left, the placenta releases exosomes carrying immuno-supportive material to the body. On the right, a diagram of an exosome showing the presented proteins on the surface on the membrane and the therapeutic cargo inside of it.
Placenta Exosomes are great! So:
Taking advantage of their induced tolerant protective environment, in this blog I will suggest a novel approach to help MS patients around the world to benefit from pregnancy, even if they can't or didn't want to carry a human being inside them for 9 months at a time. I assume that´s many of you.
Your own living pharmacy, inside you!
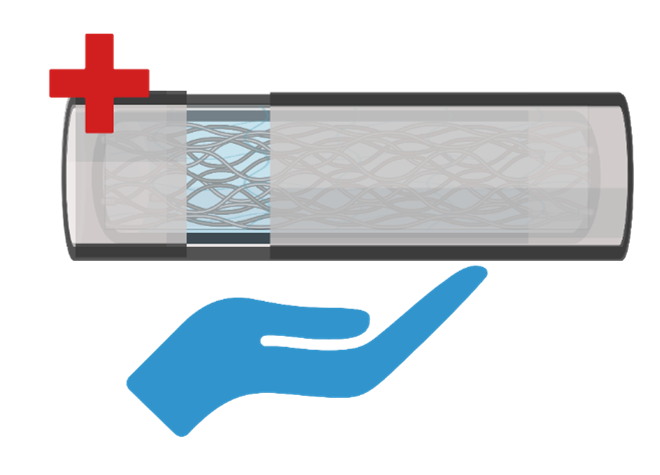
Figure 3. An implant system to deliver constant steady stream of therapeutic exosomes from placenta cells
This is pregnancy in a capsule. A permanent yet retrievable, implanted system to deliver therapeutic placenta exosomes 6 with all their superpowers in a sustained long-term manner. Inside the capsule is a mesh of hydrogel scaffolding placental stem cells harvested from the mother herself postpartum or donated from healthy unwanted placentas. The capsule is porous and breathable to allow the cells to breathe and receive nutrients, however, the pores are too small for your immune cells to get to them, only the exosomes can get out 13. These stem cells and exosomes are also bio-engineered to further strengthen their therapeutic capabilities and load them up with all the needed materials not only to replicate a pregnancy´s state of immune tolerance but to also ensure recovery and remyelination 6.
This is done by secreting a low steady dose of Interferon-beta, an anti-inflammatory and one of the first MS disease modifying drugs. The cells will also be engineered to secrete more of the peace-making PD-L1 proteins we mentioned earlier, and IL-10 to sustain anti-inflammation 15. Most importantly, they will be made to secrete all the needed miRNAs to ensure the right state of immunity. These miRNAs, usually the C19MC cluster 12, are able to silence the attack genes and help expand the protective Treg cells instead of the offensive Th1 cells.
And Exosomes are the perfect peacekeepers due to their small nano scale size and their ability to cross the blood brain barrier, where the attacks usually occur.
Your own stream of armored protective placenta exosomes traveling from under your skin all the way crossing the Blood Brain Barrier and delivering their anti-inflammatory and gene regulating cargo directly at the site of the damage. This ease of travel is also ensured by engineering the surface of the exosomes to express the rabies virus glycoprotein (RVG) peptide, a short neuron specific peptide, which is demonstrated to cross the BBB and specifically target brain cells 6.
While each of these components have been studied as a standalone solution using different mechanisms, there seems to be no such solution that takes advantage of the slow release ability of an implanted device to deliver placenta stem cells exosomes that are further engineered to ensure the effectiveness and ease of transport of all needed miRNAs and immune-factors for the modulation of the immune system and specifically for multiple sclerosis.
Figure 4. The capsule implant system with a hydrogel mesh inside of it where placenta stem cells are grown and engineered to secrete a specific type of exosomes with extra therapeutic powers for treating MS. The picture on top shows the capsule with the mesh inside of it. The picture under it shows a zoom-in perspective of the exosomes carried within the cells in the capsule.
So, what could this mean for you?
With a solution like this, you don't have to worry about sticking to a strict schedule of regular injections or long, inconvenient IV sessions. That will be a thing of the past, since you have it all built in. This “living pharmacy” is designed to be an effective sustainable therapeutic with great advantage over other solutions that only get you a short-term fix 14.
This placenta stem cell implant is the perfect combination of immunology, endocrinology, and nanotechnology working together to bring harmony back to your immune system rather than fighting against it. You can rest assured that researchers around the world are putting all their knowledge and tricks together, to make such advancements in science a reality and help millions of MS patients around the world.
Figure 5: A mother and patient with an MS happily breastfeeding her child and feeling healthy with the implant under her skin.
References:
- Joo, J. S., Lee, D., & Hong, J. Y. (2024). Multi-Layered mechanisms of immunological tolerance at the Maternal-Fetal Interface. Immune Network, 24(4). https://doi.org/10.4110/in.2024.24.e30
- Bai, K., Li, X., Zhong, J., Ng, E. H. Y., Yeung, W. S., Lee, C., & Chiu, P. C. N. (2021). Placenta-Derived exosomes as a modulator in maternal immune tolerance during pregnancy. Frontiers in Immunology, 12. https://doi.org/10.3389/fimmu.2021.671093
- Martire, S., Montarolo, F., Spadaro, M., Perga, S., Sforza, M. L., Marozio, L., Frezet, F., Bruno, S., Chiabotto, G., Deregibus, M. C., Camussi, G., Botta, G., Benedetto, C., & Bertolotto, A. (2021). A First Phenotypic and Functional Characterization of Placental Extracellular Vesicles from Women with Multiple Sclerosis. International Journal of Molecular Sciences, 22(6), 2875. https://doi.org/10.3390/ijms22062875
- Bai, K., Lee, C., Liu, X., Li, J., Cao, D., Zhang, L., Hu, D., Li, H., Hou, Y., Xu, Y., Kan, A. S. Y., Cheung, K., Ng, E. H. Y., Yeung, W. S. B., & Chiu, P. C. N. (2022). Human placental exosomes induce maternal systemic immune tolerance by reprogramming circulating monocytes. Journal of Nanobiotechnology, 20(1). https://doi.org/10.1186/s12951-022-01283-2
- Al-Shammri, S., Rawoot, P., Azizieh, F., AbuQoora, A., Hanna, M., Saminathan, T., & Raghupathy, R. (2004). Th1/Th2 cytokine patterns and clinical profiles during and after pregnancy in women with multiple sclerosis. Journal of the Neurological Sciences, 222(1–2), 21–27. https://doi.org/10.1016/j.jns.2004.03.027
- Zonouz, A. M., Rahbardar, M. G., & Alibolandi, M. (2025). Exosome-based platforms for treatment of multiple sclerosis. Brain Research Bulletin, 222, 111256. https://doi.org/10.1016/j.brainresbull.2025.111256
- Naserpour, L., Jalise, S. Z., Khoshandam, M., & Hosseinzadeh, F. (2025). Molecular mechanism and the main signaling pathways in the development and progression of Multiple Sclerosis. Experimental Cell Research, 114769. https://doi.org/10.1016/j.yexcr.2025.114769
- Rizvi, S. A., & Coyle, P. K. (2011). Clinical Neuroimmunology: Multiple Sclerosis and Related Disorders. Humana Press.
- Devvanshi, H., Kachhwaha, R., Manhswita, A., Bhatnagar, S., & Kshetrapal, P. (2022). Immunological changes in pregnancy and prospects of therapeutic PLA-Xosomes in adverse pregnancy outcomes. Frontiers in Pharmacology, 13. https://doi.org/10.3389/fphar.2022.895254
- Sabapatha, A., Gercel‐Taylor, C., & Taylor, D. D. (2006). Specific Isolation of Placenta‐Derived Exosomes from the Circulation of Pregnant Women and Their Immunoregulatory Consequences1. American Journal of Reproductive Immunology, 56(5–6), 345–355. https://doi.org/10.1111/j.1600-0897.2006.00435.x
- Favaro, R. R., Murrieta‐Coxca, J. M., Gutiérrez‐Samudio, R. N., Morales‐Prieto, D. M., & Markert, U. R. (2020). Immunomodulatory properties of extracellular vesicles in the dialogue between placental and immune cells. American Journal of Reproductive Immunology, 85(2). https://doi.org/10.1111/aji.13383
- Ouyang, Y., Bayer, A., Chu, T., Tyurin, V. A., Kagan, V. E., Morelli, A. E., Coyne, C. B., & Sadovsky, Y. (2016). Isolation of human trophoblastic extracellular vesicles and characterization of their cargo and antiviral activity. Placenta, 47, 86–95. https://doi.org/10.1016/j.placenta.2016.09.008
- Lathuilière, A., Mach, N., & Schneider, B. (2015). Encapsulated cellular implants for recombinant protein delivery and therapeutic modulation of the immune system. International Journal of Molecular Sciences, 16(5), 10578–10600. https://doi.org/10.3390/ijms160510578
- Nahayati, M. A., Yazdi, S. G., Hassanisabzevar, M., Baniasadi, P., Forouzanfar, F., & Saburi, E. (2025). Mesenchymal Stem Cells in Clinical Trials for Multiple Sclerosis: A Systematic Literature Review and review of clinical trials. Multiple Sclerosis and Related Disorders, 106694. https://doi.org/10.1016/j.msard.2025.106694
- Clark, K. C., Wang, D., Kumar, P., Mor, S., Kulubya, E., Lazar, S. V., & Wang, A. (2022). The molecular mechanisms through which placental mesenchymal stem Cell‐Derived extracellular vesicles promote myelin regeneration. Advanced Biology, 6(2). https://doi.org/10.1002/adbi.202101099
- Bernardi, M. D. L., Rus, N., Vernooij, R. W. M., Verhaar, M. C., & Rookmaaker, M. B. (2025). Cell macroencapsulation devices in contemporary research: A systematic review. Regenerative Therapy, 30, 144–156. https://doi.org/10.1016/j.reth.2025.05.013
- Burkova, E. E., Sedykh, S. E., & Nevinsky, G. A. (2021). Human placenta exosomes: biogenesis, isolation, composition, and prospects for use in diagnostics. International Journal of Molecular Sciences, 22(4), 2158. https://doi.org/10.3390/ijms22042158
- López-Muguruza, E., Villar-Gómez, N., Matias-Guiu, J. A., Selma-Calvo, B., Moreno-Jiménez, L., Sancho-Bielsa, F., Lopez-Carbonero, J., Benito-Martín, M. S., García-Flores, S., Bonel-García, N., Kamal, O. M., Ojeda-Hernández, D., Matías-Guiu, J., & Gómez-Pinedo, U. (2022). The integration of cell therapy and biomaterials as treatment strategies for remyelination. Life, 12(4), 474. https://doi.org/10.3390/life12040474
- Magill, E., Demartis, S., Gavini, E., Permana, A. D., Thakur, R. R. S., Adrianto, M. F., Waite, D., Glover, K., Picco, C. J., Korelidou, A., Detamornrat, U., Vora, L. K., Li, L., Anjani, Q. K., Donnelly, R. F., Domínguez-Robles, J., & Larrañeta, E. (2023). Solid implantable devices for sustained drug delivery. Advanced Drug Delivery Reviews, 199, 114950. https://doi.org/10.1016/j.addr.2023.114950
- Wang, X., & Li, D. (2020). The mechanisms by which trophoblast-derived molecules induce maternal–fetal immune tolerance. Cellular and Molecular Immunology, 17(11), 1204–1207. https://doi.org/10.1038/s41423-020-0460-5




Add comment
Comments
This was a really interesting read. I’ve heard before that MS symptoms often ease up during pregnancy, but I never knew how complex the immune side of it actually is. The explanation about the placenta using exosomes to calm the immune system made a lot of sense. It’s a clever way to describe something that sounds incredibly intricate.
The idea of a “pregnancy in a capsule” is pretty fascinating. Turning that natural tolerance process into a steady, controlled therapy feels like a logical next step for autoimmune research. I’m curious how consistent exosome production would be over time, and whether the implant would need maintenance or replacement once the cells inside slow down.
It also made me wonder if the same concept could apply to other autoimmune conditions, since a lot of them involve a similar kind of immune imbalance.
Really thoughtful and creative concept! It’s exciting to see this kind of cross between immunology and bioengineering being explored.
Hi Sarcha,
Thank you for your comment and taking the time to read my blog!
You're right in wondering about the long-term performance and maintenance of such a device as it is rather invasive of a solution and one wouldn't want to go through putting a foreign body under their skin every couple of months or weeks.
I imagine we'd have to look into the proliferative capacity and longevity of these cells and bio-engineering them to create the specific exosomes with the needed cargo and increase their life span, perhaps through hybridization or genetic modification to delay cellular senescence or by co-encapsulation with supportive biomaterials to enhance viability.
But that still won't change the fact that the patients would probably still have consistent monitoring every year or so and maybe plan an exchange every few years depending on the output performance and the degree of scarring if it exists.
As this idea is immunomodulatory in its nature, I don't see why it shouldn't work for other autoimmune diseases like Rheumatoid Arthritis or Systemic Lupus Erythematosus for example.
We know that cell encapsulation is a proven delivery method and we know that using exosomes as therapeutic cargo also works as research has shown before, but this idea of merging the strong immunomodulatory aspects of placental cell exosomes and long term implantation in this form I suggest is a new approach (as far as I know, especially for MS) and much remains to be learned about exosome production consistency and optimal delivery here.
Wait, this is actually a really cool idea. How'd you come up with this? This is wild.
I'm curious, is there a version of this that could be done for a broader range of patients or would this be locked to postpartum mothers as a treatment?
Hi Simon,
I´m really glad you liked the proposal!
I actually started with a sci-fi idea of “what if we could somehow implant a fake embryo that keeps that protection going for longer”, of course i quickly realized that´s ridiculous!
But then it took me to thinking about implant IUDs and placenta mesenchymal stem cells. I wondered if one could create such an implant but with what makes a placenta instead of hormones and then started reading about cell encapsulation strategies.
I realized no one had tried creating this sort of implants for Multiple sclerosis treatment in this way, there was some work done on implants of stem cells directly at demyelinated cells to help them recover but that's different as it aims to heal and it uses a different implantation technique and cell lines with the aim of remyelination not to protect.
As I see it, it should be a viable option for everyone as the device doesn't pump out hormones but exosomes with cytokines and mRNAs to reset the inflammation.
Of course this would have to be tested and there could be ways that these extra mRNAs go to the wrong place causing damage rather than helping because we know that the same mRNA doing good in one place could cause havoc somewhere else.
Have you thought much about how this could potentially bring about adverse effects associated with pregnancy hormones, such as mood swings, and nausea? Also, how would this affect physiological changes that normally return to normal postpartum and after the breastfeeding stage, for example, Breast development and the menstrual cycle?
Sorry about this. I was pasting my comment in from a word document and i accidentally posted only the final question! Heres the full comment:
Wow! I know we were supposed to find a novel solution but this is a real "bleeding edge" concept. It really gives an insight into the pathways we could be looking at to treat non-communicable, and particularly autoimmune diseases. The idea that IV treatment could become redundant is something that would reap huge rewards for improving the quality of life for patients. It would certainly beneficial to the mental health of mothers with MS, who already have enough to worry about.
Have you thought much about how this could potentially bring about adverse effects associated with pregnancy hormones, such as mood swings, and nausea? Also, how would this affect physiological changes that normally return to normal postpartum and after the breastfeeding stage, for example, Breast development and the menstrual cycle?
Hi Dallan! I really appreciate your comment and I'm glad you liked the proposal.
The idea so far is that we don't change anything related to hormones and only focus on the placenta exosomes with special therapeutic cargo that acts as an immunomodulatory solution. Otherwise, I agree with you that changing these hormones does have the risk of causing unwanted reactions and problems.
As long as it's only cytokines and mRNA targeting inflammation, I hope to achieve a successful and stable therapy in all stages of a person´s life, whether pregnant or not.
Thank you for taking the time to read and leave a comment
This idea sounds AMAZING! It sounds like such a great solution to the problem at hand. The language you used in describing the mechanism ex describing the exosomes as peacekeepers made the piece quite fun to read too. I wanted to ask, which part of the body's skin would you be placing it in?
Hey Baneen! Happy you like the idea :)
That's a very good question. Honestly, I think we’d have to consider factors like blood flow and skin thickness to avoid irregular release rates or serious scarring. The upper arm might be suitable mainly because it’s easy to access and handle, plus the aesthetic impact of any scarring could be minimized by placing it in a less visible area.
But we could for sure benefit from looking into other options like the thigh, back, or stomach
I’ve never heard anything like this before! The idea placental exosomes could be used as a treatment for MS is really exciting. It’s amazing to think that something the body naturally does could inspire such a futuristic therapy an advanced therapy.