Introduction
In Ireland, we are heavily connected with sport, either through family and friends or as a player. Some main sports played across Ireland include GAA, rugby and boxing, which are all high impact. Did you know that a sports related concussion or impact to your head can lead to a traumatic brain injury (TBI)? And that repeated TBIs can increase your likelihood of developing Chronic Traumatic Encephalopathy (CTE). (1) In reported CTE cases, over 97% have been associated with repetitive head impacts, which are seen periodically in contact sports.
What is CTE and its symptoms?
CTE’s symptoms are delayed and progress over time, following earlier minor brain traumas. There has been a lot of research into CTE but ultimately, there is no current treatment.
Part 1 of Novel treatment: Inhibiting cell death and stimulate cell regrowth in CTE
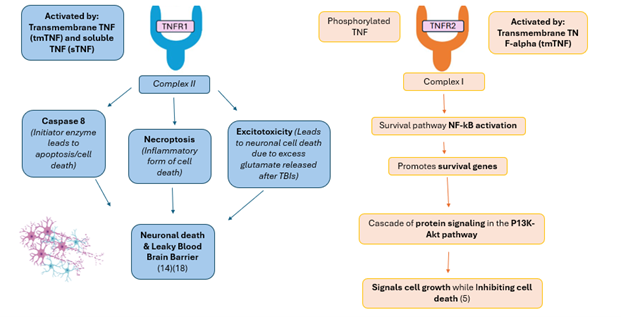
In mechanical trauma such a Traumatic Brain Injuries, the brain’s glial cells (non-neuronal cells) release TNF- α (pro-inflammatory cytokine small protein) to the site of injury. Unfortunately, repetitive TBI’s such as those seen in impact sports, can lead to the development of CTE. During immune activation, glial cells in the Central Nervous System release TNF-α cytokine, to initiate TNFR#1 and TNFR2 response. The issue in CTE is that excessive amounts of TNF- α can lead to neuron excitotoxity as well, as self-reactivation, leading to further amounts of TNF-alpha being produced. (2)(3) Many clinically approved treatments, known as anti-TNF drugs, can suppress this response by blocking the over expressed TNF- α. However, by doing so, it also inhibits the beneficial activation of TNFR2 which can stimulate neurogenesis (neuron renewal). (4)
Potential TNF-alpha initiated pathways
Approach
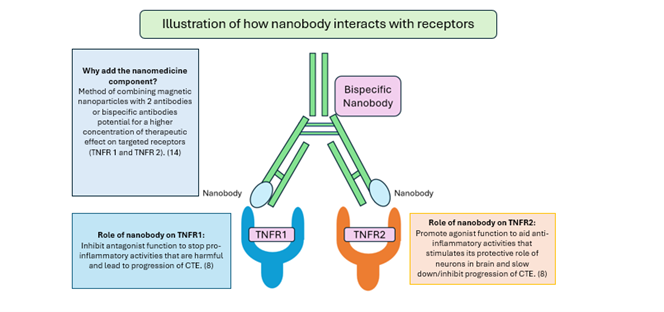
The suggested novel drug must show high affinity to each target receptor to compete with and overcome, the naturally high levels of TNF-α in patients suffering with CTE. The weakened blood brain barrier allows for easier transport to its target, combined with a suggested, nanoparticle targeted delivery. By adding the nanoparticle component to a bispecific antibody, this will facilitate the above requirements with an effective therapeutic effect.
Tissue specific responses must be researched to ensure TNFR2 activation, promotes its agonist effects. Clinical trials will need to be initiated to support the effect of TNFR1 and TNFR2 on different areas. (6)
Anti-TNF-α drugs have been researched for other diseases and has shown promising effects on TNFR1 and TNFR2 inhibition. This research on anti-TNF-α therapies in other diseases include Crohn's disease and Rheumatoid arthritis. By combining this approach, it can target excess TNF-α, which reduces Chronic neuroinflammation. (7) However, this also prevents TNFR2 activation, which has beneficial functions that we would like to promote in our novel treatment. See functions of both TNFR1 and TNFR2 in table below.
Dual Treatment
By manipulating a dual treatment method using, Step 1: anti-TNF-α drugs with, Step 2: our proposed bispecific nanobody which functions on TNF-α, absence. This could supress the effects of chronic neuroinflammation, while selectively inhibiting TNFR1 activation and promoting TNFR2 stimulation. We would hope to see a reduction in the TNF cell death signalling blockade and an enhancement of neural growth with little side effects. (10)
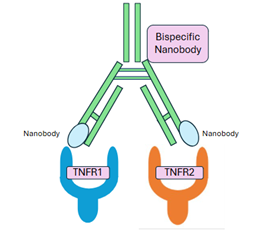
Step 2: Bispecific Nanobody
Part 2 of Novel treatment: Targeting TNFR1 and TNFR2 (Receptors) with Bispecific Nanobodies
What are Bispecific Nanobodies?
-
Bispecific antibodies have been largely studied over the past decade to target/bind to specific antigens (can generate an immune response), to improve the effectiveness of a treatment against a particular disease. (11).
-
In the context of nanomedicine, nanoparticle-based antibodies have been used to trigger structural changes to targeted area which can reduce its stability and allow for targeted drug delivery. Additionally, these nanotechnology-based antibodies have been used to facilitate the release of a drug at a specific target tissue. (12)
Nanobodies targeting TNRF 1 and TNFR 2
-
Previous research has shown that nanobodies have promoted the agonist (anti-inflammatory) role of TNFR2, whilst also promoting the antagonist (inflammatory) role of TNFR1.
-
With our proposed dual treatment, the anti-TNF-α drug would help reduce and counteract the harmful inflammatory TNFR1 activation that trigger’s cell death.
-
The bispecific nanobody will then stimulate different signalling pathways, which will lead to the beneficial anti-inflammatory effects, helping relieve symptoms of a CTE patient. (13)
For our novel treatment, both the anti-TNF-α drug and bispecific nanobodies will be used to target both receptors TNFR1 and TNFR2 which would be delivered intravenously (via the vein) to patient. (14) See illustration below for how the Bispecific nanobody solely reacts with TNFR1 and TNFR2.
Conclusion
In conclusion, the novel treatment of using an anti-TNF-α drug, followed by a bispecific nanobody to reduce inflammation (TNFR1) and to promote its anti-inflammatory functions (TNFR2), with the enhanced therapeutic effect from the antibody nanoparticle component. This approach could be analysed for its effectives across multiple inflammatory diseases and hopefully have life changing impacts. Chronic Traumatic Encephalopathy CTE could affect any of us from playing the sports we love, it is so important we continue to research potential novel treatments!
References
- Lucke-Wold, B. P., Turner, R. C., Logsdon, A. F., Bailes, J. E., Huber, J. D., & Rosen, C. L. (2014). Linking traumatic brain injury to chronic traumatic encephalopathy: identification of potential mechanisms leading to neurofibrillary tangle development. Journal of neurotrauma, 31(13), 1129–1138. https://doi.org/10.1089/neu.2013.3303
- Mayo Clinic. (2023). Chronic traumatic encephalopathy - symptoms and causes. Mayo Clinic. https://www.mayoclinic.org/diseases-conditions/chronic-traumatic-encephalopathy/symptoms-causes/syc-20370921
- García-Domínguez, M. (2025). The Role of TNF-α in Neuropathic Pain: An Immunotherapeutic Perspective. Life, 15(5), 785. https://doi.org/10.3390/life15050785
- Richter, F., Zettlitz, K. A., Seifert, O., Herrmann, A., Scheurich, P., Pfizenmaier, K., & Kontermann, R. E. (2019). Monovalent TNF receptor 1-selective antibody with improved affinity and neutralizing activity. mAbs, 11(1), 166–177. https://doi.org/10.1080/19420862.2018.1524664
- Faustman, D. L., Davis, M., & Kuhtreiber, W. M. (2025). TNFR2 Agonism: Basic Science and Promising Treatment for Multiple Sclerosis and Related Diseases. International Journal of Molecular Sciences, 26(16), 7839. https://doi.org/10.3390/ijms26167839
- Chen, Y., Jiang, M., & Chen, X. (2023). Therapeutic potential of TNFR2 agonists: a mechanistic perspective. Frontiers in immunology, 14, 1209188. https://doi.org/10.3389/fimmu.2023.1209188
- Li, Y., Ye, R., Dai, H., Lin, J., Cheng, Y., Zhou, Y., & Lu, Y. (2025). Exploring TNFR1: from discovery to targeted therapy development. Journal of translational medicine, 23(1), 71. https://doi.org/10.1186/s12967-025-06122-0
- Fiedler, T., Fairless, R., Pichi, K. et al.Co-modulation of TNFR1 and TNFR2 in an animal model of multiple sclerosis. J Neuroinflammation 20, 100 (2023). https://doi.org/10.1186/s12974-023-02784-z
- Lotocki, G., Alonso, O. F., Dietrich, W. D., & Keane, R. W. (2004). Tumor necrosis factor receptor 1 and its signaling intermediates are recruited to lipid rafts in the traumatized brain. The Journal of neuroscience : the official journal of the Society for Neuroscience, 24(49), 11010–11016. https://doi.org/10.1523/JNEUROSCI.3823-04.2004
- Gonzalez Caldito N. (2023). Role of tumor necrosis factor-alpha in the central nervous system: a focus on autoimmune disorders. Frontiers in immunology, 14, 1213448. https://doi.org/10.3389/fimmu.2023.1213448
- Sun, Y., Yu, X., Wang, X., Yuan, K., Wang, G., Hu, L., Zhang, G., Pei, W., Wang, L., Sun, C., & Yang, P. (2023). Bispecific antibodies in cancer therapy: Target selection and regulatory requirements. Acta pharmaceutica Sinica. B, 13(9), 3583–3597. https://doi.org/10.1016/j.apsb.2023.05.023
- Hillman, Y., Lustiger, D., & Wine, Y. (2019). Antibody-based nanotechnology. Nanotechnology, 30(28), 282001. https://doi.org/10.1088/1361-6528/ab12f4
- Anany, M. A., Siegmund, D., Zaitseva, O., Lang, I., Mouhandes, B., Salah, M., Weisenberger, D., Stepanzow, S., Vargas, J. G., Dahlhoff, J., Schneider, T., Tanghe, G., Stevenaert, F., Tezil, T., Van Belle, T., Kar, B., Dandekar, T., Hermanns, H. M., van Rompaey, L., Beilhack, A., … Wajant, H. (2025). Valence and avidity determine the agonistic activity of anti-TNFR2 nanobody fusion proteins. Cell chemical biology, 32(10), 1279–1292.e6. https://doi.org/10.1016/j.chembiol.2025.09.002
- Tang, J., Shen, D., Zhang, J., Ligler, F. S., & Cheng, K. (2015). Bispecific antibodies, nanoparticles and cells: bringing the right cells to get the job done. Expert opinion on biological therapy, 15(9), 1251–1255. https://doi.org/10.1517/14712598.2015.1049944
- Caplacizumab. (2021, June 7). Nih.gov; National Institute of Diabetes and Digestive and Kidney Diseases. https://www.ncbi.nlm.nih.gov/books/NBK571859/






Add comment
Comments
This is a really interesting idea for a treatment. I like the two step approach. I have one question about getting the drug into the brain. You mention the barrier that protects the brain is weaker in CTE patients. Since CTE symptoms show up years after the hits to the head, how can you be sure that this barrier is still weak enough for the drug to get through when you give the treatment? Would you need a way to temporarily open this barrier to make sure the medicine works?
Hi Robyn,
Thanks so much for your question! It is important to highlight that TNF-Alpha is key marker of Traumatic Brain Injury TBI, but it is also a leading indicator of Chronic Traumatic Encephalopathy CTE. (Bindra et al. 2025) In patients with CTE, they will have naturally higher levels of TNF-alpha which increases the blood brain barrier permeability. Therefore, the blood brain barrier would be weak enough to allow the medicine to work!
But with the first part of this dual approach being an anti-TNF-alpha drug, we could question if this would inhibit or reduce the effects of the second part being a bispecific antibody to target TNFR1 and TNFR2 in the brain. TNF-Alpha is not the only regulator in increasing the Blood brain barrier permeability. Other elements will still play a role in its permeability in CTE patients such as pro-inflammatory cytokines e.g. Interleukin-1β. In a study, it was found that the impact of both TNF-alpha and interlukin-1β will affect the blood brain barrier permeability via different mechanisms. Interleukin-1β and other pro-inflammatory cytokines would be able to solely maintain the permeability of the blood brain barrier to allow the bispecific nanobody to carry out its mechanism of action of TNFR1 and TNFR2. (Versele et al. 2022)
Overall, to answer your question, we wouldn't need a way to temporarily open the barrier due to the natural pro-inflammatory cytokines in CTE patients keeping this barrier weakened to allow our drugs to take action!
Bindra, G.S., Asad, S., Shanaa, J., Lui, F., Budson, A.E., Turk, K.W. and Cherry, J.D., 2025. Neuroinflammatory mechanisms may help identify candidate biomarkers in chronic traumatic encephalopathy (CTE). Free Neuropathology, 6, p.15.
Versele, R., Sevin, E., Gosselet, F., Fenart, L. and Candela, P., 2022. TNF-α and IL-1β modulate blood-brain barrier permeability and decrease amyloid-β peptide efflux in a human blood-brain barrier model. International Journal of Molecular Sciences, 23(18), p.10235.
Hi, Given your proposed treatment, would the anti-TNF-α drug have any inhibiting effects on your bispecific nanobody? Is this something that you can overcome?
Hi Elaine, great question, thank you for reaching out!
Our synergistic approach offers a high level overview of the therapeutic design. However, based on your query, it is important to mention that the nanobodies variable domain were carefully engineered with opposing binding site affinities. Any indirect binding, is overcome by paratope specific affinity maturation, to ensure the highest affinity for each paratope's target epitope. This ensures that anti-TNF-alphas mitigating effects on normal levels of TNF-alpha in the body are achieved, while inhibiting competition for our proposed bispecific nanobody.