
With more than 120 linked deaths due to adverse effects there is a great need for improvement of Post-Finasteride Syndrome (PFS) therapy1. The drug - Finasteride - to which this syndrome is linked is normally prescribed to men suffering from androgenic alopecia to aid in hair growth. But what started with opening the pill container to fix a receding hairline can turn into opening Pandora’s Box. Upon discontinuation, there is a plethora of adverse effects which patients can suffer from, decreasing their life’s quality far beyond the point which hair loss could’ve pushed them to2. This blog post is written with the intent to highlight how leveraging the gut microbiome can aid in relieving PFS symptoms and give you as reader a deeper understanding how the microbiome influences how we feel.
PFS’s poorly defined status and some of its symptoms
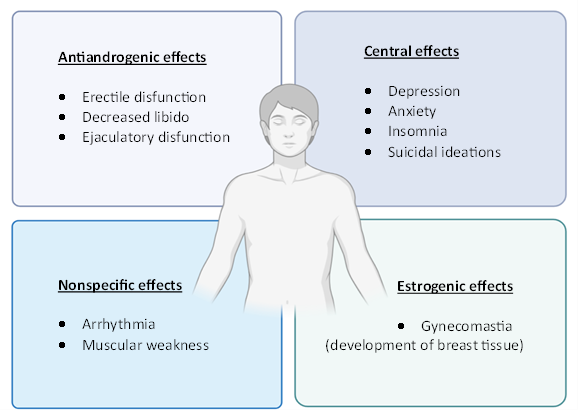
Diagnosing a patient with PFS has been quite difficult since to this day no medical society has proposed a clear definition for it3. Because of this, people who end up developing serious symptoms after discontinuation of Finasteride are often left undiagnosed and without further guidelines on what to do. Upon collection of the patients reported symptoms, they can be grouped into four clusters - antiandrogenic, estrogenic, central and unspecific effects. A person affected by PFS can exhibit symptoms from only one of these categories or display symptoms from multiple categories with a persistence time of these symptoms from weeks to multiple years3.
Figure 1: Display of the four symptom clusters with examples for how the effects manifest in PFS patients.
Finasteride’s mechanisms of action including steroid alterations
Most of the above-mentioned symptoms can be linked to a dysregulation of the finely tuned system of steroid hormones - the exact area where Finasteride is creating a disbalance. It selectively inhibits an enzyme, 5-alpha-reductase (5aR), that is responsible for the conversion of testosterone into dihydrotestosterone (DHT)4. Finasteride mainly affects isoform 2 of 5aR which is mostly found in the prostate, testicles, hair follicles and in the liver5. The inhibition if the testosterone conversion leads to a decreased amount of DHT in the periphery – which then in turn results in a decreased binding of DHT to androgen receptors (AR). Since the AR activation through DHT results in ten times higher signaling intensities compared to testosterone, the decreased DHT levels alter important signaling pathways5. Another important consideration in therapeutical approaches is that the body needs almost a full month for half of the inhibited 5aR isoform 2 enzymes to be degraded4. After discontinuation of Finasteride a study has shown that DHT levels remain low in PFS patients together with other neuroactive steroids like progesterone and its metabolites like allopregnanolone6. In keeping with the theme of metabolites and steroid hormones I want to propose a gut microbiome focused therapy to improve PFS patients’ symptoms.
The gut-brain-axis and how the bacteria in our guts influences our health
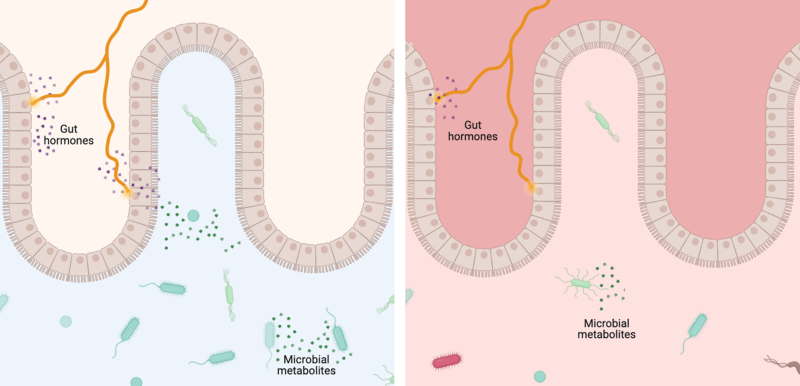
The bacteria colonizing the gut of a healthy human creates a plethora of metabolites which can be taken up by the cells in our colon and have a direct influence on that human’s mood and bodily functions. PFS patients have shown a dysregulation in their gut microbiome with several bacterial species being underrepresented after the Finasteride therapy - for example Faecalibacterium and Ruminococcaceae species6,7. The decrease in bacterial species leads to a deficiency of their metabolites which have a protective effect for the host (aka the human). Short-chain fatty acids (SCFA) like butyrate and propionate belong to these metabolites and have been linked to sleep modulation, alleviation of depressive symptoms and anti-inflammatory effects8. The lack of these can be clearly linked to the PFS symptomatology.
Figure 2: Normal gut microbiome (left) with healthy bacterial growth supplying the body with metabolites and gut hormones versus abnormal gut microbiome (right) with decreased healthy bacterial growth and fewer gut metabolites.
For a therapy approach I would propose a probiotic supplementation with bacterial strains which are producers of SCFA or - for severe dysbiosis cases - a stool transplant from a healthy individual. If the beneficial bacteria can be brought back to the colon of the person suffering from PFS the metabolites could aid in alleviating the symptoms. The gut microbiome can also be described as an endocrine organ because beside SCFA the microbiome is also able to metabolize steroid hormones9. For PFS patients it is especially interesting that some microbes have a significant amount of 5aR isotype 1 enzymes which could support the testosterone to DHT conversion. Since DHT cannot be converted into estrogens anymore after the conversion by 5aR this could be especially interesting for PFS patients suffering from estrogenic effects plus it has the benefit of increasing DHT levels during early stages after the Finasteride therapy was stopped. Two bacterial strains shown to have 5aR isotype 1 gene expression are Odoribacter splanchnicus and Parabacteroides distasonis which should be added to the list of probiotics to be considered9. Another final thought is that through alteration of the bacterial strains in our gut the total amount of serotonin, the “feel-good” hormone, is changed since 95% of the human body’s serotonin is produced in the gut. With the goal to increase the number of bacteria which produce serotonin, probiotic treatment with Bifidobacterium and Lactobacillus strains could be of interest to lower anxiety and improve depressive symptomatology8.
Figure 3: The addition of the outlined bacterial strains (left) could make a difference in gut health and should result in an increase of beneficial metabolites for the brain
This proposed therapy would leverage all the positive effects of beneficial gut bacteria to improve the patients’ mental status and gut health in general.
To the people who are still considering whether to start Finasteride treatment
The last paragraph is dedicated to the people that could still be dissuaded from starting a Finasteride therapy – or to at least take a few precautions before starting the medication.
In individuals who have androgenic alopecia the chance of mental disorders is higher in comparison to a non-affected control group3. By exposing yourself to the effects of Finasteride, it could lead to the development of something worse than a receding hairline. If you are set on Finasteride, maybe instead of the oral Finasteride a topical Finasteride could be chosen to have a less systemic effect. And last but not least - fueling your body with vitamin-dense and fiber-rich foods will never not be beneficial for you and for your gut bacteria.

Sources:
1 https://www.pfsfoundation.org/ (last access on 31.10.2025)
2 https://www.pfsnetwork.org/ (last access on 31.10.2025)
3 Cilio, S., Tsampoukas, G., Morgado, A., Ramos, P., & Minhas, S. (2025). Post-finasteride syndrome - a true clinical entity?. International journal of impotence research, 37(6), 426–435. https://doi.org/10.1038/s41443-025-01025-6
4 PROPECIAÒ label from latest revision 07/2022 https://www.accessdata.fda.gov/drugsatfda_docs/label/2022/020788s030lbl.pdf
5 Batista, R. L., & Mendonca, B. B. (2020). Integrative and Analytical Review of the 5-Alpha-Reductase Type 2 Deficiency Worldwide. The application of clinical genetics, 13, 83–96. https://doi.org/10.2147/TACG.S198178
6 Diviccaro, S., Oleari, R., Amoruso, F., Fontana, F., Cioffi, L., Chrostek, G., Abenante, V., Troisi, J., Cariboni, A., Giatti, S., & Melcangi, R. C. (2025). Exploration of the Possible Relationships Between Gut and Hypothalamic Inflammation and Allopregnanolone: Preclinical Findings in a Post-Finasteride Rat Model. Biomolecules, 15(7), 1044. https://doi.org/10.3390/biom15071044
7 Giatti, S., Diviccaro, S., Cioffi, L., & Cosimo Melcangi, R. (2024). Post-Finasteride Syndrome And Post-Ssri Sexual Dysfunction: Two Clinical Conditions Apparently Distant, But Very Close. Frontiers in neuroendocrinology, 72, 101114. https://doi.org/10.1016/j.yfrne.2023.101114
8 Zhu, Z., Cheng, Y., Liu, X., Xu, X., Ding, W., Ling, Z., Liu, J., & Cai, G. (2025). The microbiota-gut-brain axis in depression: unraveling the relationships and therapeutic opportunities. Frontiers in immunology, 16, 1644160. https://doi.org/10.3389/fimmu.2025.1644160
9 Ohlsson, C., Li, L., Horkeby, K., Lawenius, L., Colldén, H., Sjögren, K., Baldanzi, G., Engström, G., Ärnlöv, J., Orho-Melander, M., Fall, T., & Grahnemo, L. (2025). The circulating dihydrotestosterone/testosterone ratio is increased by gut microbial 5α-reductase activity in females. EBioMedicine, 121, 105978. Advance online publication. https://doi.org/10.1016/j.ebiom.2025.105978



Add comment
Comments
I think the focus on restoring a healthy gut microbiome is really interesting and seems like a good idea to help symptoms as the gut is so important for our overal health. One question I think it would be important to investigate is whether the microbiome changes are a cause of PFS symptoms or more of a side effect of Finasteride. Understanding this could help refine the therapy approach
Super interesting! Yet another example of how the gut-brain axis could impact a neurological disease. Although, Jack definitely has a point, it would require a dedicated study to determine the order of events on whether the dys-regulated microbiome comes before or after the PFS.