Problem: when treating hair loss leads to neurological disorders
Finasteride is a common prescription medicine to treat hair loss and prostate enlargement; however, some people experience long-term symptoms in sexual, psychiatric and physical after stopping the drug. This condition is called Post-Finasteride Syndrome (PFS), which can be devastating but does not happen to all finasteride users (PFS Foundation, n.d).
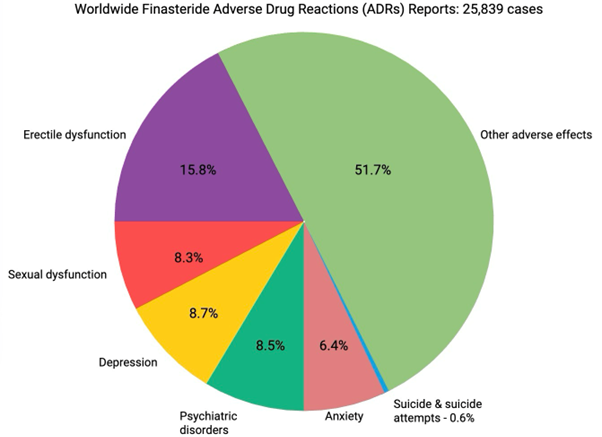
According to the PFS Foundation (n.d), there are more than 25000 reports of side effects from who had taken this medicine, in which 4,083 cases of erectile dysfunction, 2,237 of depression, 2,155 of other sexual dysfunction, 1,644 of anxiety, 2,189 of other psychiatric disorders, 121 deaths by suicide, and 54 suicide attempts. Figure 1 illustrates the percentage of these adverse drug reactions to this drug out of the total number of global records.
Figure 1: Distribution of Adverse Drug Reactions (ADRs) Reports related to finasteride (PFS Foundation n.d) (Created by using Biorender)
Pathophysiology: Disruption of synthesis neurosteroid
Finasteride helps to prevent testosterone from converting to dihydrotestorone (DHT) – the hormone that causes hair loss by inhibiting the enzyme 5-alpha reductase. When DHT is reduced, hair loss is reduced. However, this enzyme also involves in synthesising neurosteroids such as allopregnanolone which regulates mood and nerve function. When allopregnanolone level drops low, users might experience serious neurological and psychological effects.
This is supported by research of Zhang et al. (2022) on different patients between 20 to 28 ages suffered persistent symptoms in neuro and endocrine after 1.5 to 2 years using finasteride and cessation times. From clinical findings, the authors suggests that these abnormal might be related to trouble in neurosteroid metabolism, especially allopregnanolone.
Recommendation: Palmitoylethanolamide (PEA) treatment
Palmitoylethanolamide (PEA) is a natural fatty compound found in our body and foods such as eggs and peanuts. It has ability to enhance allopregnanolone synthesis process, reduce neuroinflammation and improve behaviour of anxiety and cognition. Pathological mechanism and treatment pathway of PFS and PEA is described in Figure 2.
Figure 2. Pathophysiology and Treatment (Created by using Biorender)
A study of Sasso et al. (2012) proved that pain relieving effect of PEA was dependent on the increase of allopregnanolone in brain of rats with chronic pain, while this neurosteroid was blocked, efficiency of PEA was lost. In addition, a report by Petrosino & Di Marzo (2017) showed that PEA reduces neuroinflammation and stabilises immune cell activity in the brain. Therefore, it helps to improve anxiety, depression and chronic pain through PPAR- α and endocannabinoid system - a complex network in our body to maintain nerve. Clinically, a trial at Navy Medical Center San Diego (2024) is evaluating the effect of ultramicronized PEA with dosage 600mg twice a day within 8 weeks to treat chronic inflammatory pain, this dosing has been approved for safety in phase 1 by FSD Pharma.
Thereby, based on mentioned research and safety results, a treatment recommendation is as follow in table 1.
Table 1: Ingredient detail of recommended treatment (Created by using Biorender)
Conclusion
Current solutions only provide symptom relief, not a cure. PFS is still not officially recognised as a disease, due to the lack of diagnostic criteria and controlled studies, even with numerous records about serve symptoms. To move forward, clear clinical research is needed to help define PFS as a pathological entity, which is not just a complex condition. However, PEA has raised as a potential candidate due to its neurosteroid restore and protection. Further research into PEA is essential which not only opens up a new hope for PFS patient, but also targets to the root cause of this syndrome.
References:
FSD Pharma (2020) ‘Phase 1 Safety and Tolerability Study of Ultramicronized PEA’, Pharmaceutical Daily, [online], available at: https://pharmaceuticaldaily.com/fsd-pharma-reports-favorable-topline-results-from-phase-1-first-in-human-safety-and-tolerability-study-of-ultramicronized-pea/ [accessed: 30 Oct. 2025].
Navy Medical Center San Diego (2024) ‘Palmitoylethanolamide 600mg twice a day in Chronic Pain’, ClinicalTrials.gov, [online], available at: https://ichgcp.net/clinical-trials-registry/NCT06273462 [accessed: 30 Oct. 2025].
Petrosino, S. and Di Marzo, V. (2017) ‘The pharmacology of palmitoylethanolamide and first data on the therapeutic efficacy of some of its new formulations’, British Journal of Pharmacology, 174(11), pp. 1349–1365, available at: https://bpspubs.onlinelibrary.wiley.com/doi/10.1111/bph.13580 [accessed: 30 Oct. 2025].
Sasso, O., Russo, R., Vitiello, S., Mattace Raso, G., D’Agostino, G., Iacono, A., La Rana, G., Vallée, M., Cuzzocrea, S., Piazza, P.-V., Meli, R. and Calignano, A. (2012) ‘Implication of allopregnanolone in the antinociceptive effect of N-palmitoylethanolamide in acute or persistent pain’, Pain, 153(1), pp. 33–41, available at: https://www.researchgate.net/publication/51617550_Implication_of_allopregnanolone_in_the_antinociceptive_effect_of_N-palmitoylethanolamide_in_acute_or_persistent_pain [accessed: 30 Oct. 2025].
Zhang, Y., Liu, Y., Zhang, H., Zhang, Y. and Zhang, X. (2022) ‘Case report: a study of the clinical characteristics and genetic variants of post-finasteride syndrome patients’, Translational Andrology and Urology, 11(10), pp.1391–1398, available at: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC9641066/ [accessed: 19 Oct 2025].




Add comment
Comments
This post makes an interesting case for using PEA to enhance allopregnanolone by activating the PPAR-α receptor. I would be interested in discussing what enzymes are affected and how this leads to allopregnanolone synthesis.
A study conducted by Melcangi et al found after analysing methylation patterns on the genes that encode for the 5α-reductase enzyme, the SRD5A2 promoter gene was more frequently methylated in the CSF of PFS patients compared to controls. As SRD5A2 encodes for the type II isoenzyme, it is suggested that the methylation-induced silencing of SRD5A2 in the CNS could lead to persistent reductions in neuroactive steroids. Could this effect the efficacy of this treatment? Or is the activation of the PPAR-α receptor upregulating different enzymes responsible for allopregnanolone synthesis?
Reference for this paper here:
Melcangi, R.C., Casarini, L., Marino, M., Santi, D., Sperduti, S., Giatti, S., Diviccaro, S., Grimoldi, M., Caruso, D., Cavaletti, G. and Simoni, M. (2019) ‘Altered methylation pattern of the SRD5A2 gene in the cerebrospinal fluid of post-finasteride patients: a pilot study’, Endocrine Connections, 8(8), pp. 1118–1125. doi: 10.1530/EC-19-0199.
Hi, thank you for sharing a new perspective on PEA with specific evidence through the article by Melcangi et al! According to the article, SRD5A2 methylation in PFS patients may reduce the activity of 5α-reductase type II, affecting the synthesis of allopregnanolone. However, in a study by Locci and Pinna (2019), especially in Figure 5, it is shown that PEA activates PPAR-α which increases the expression of enzymes that convert cholesterol to PE (pregnenonlone - precursor of allopregnanolone) such as 5α-reductase type I (5α-RI) (or SRD5A1) and 3α-HSD. Therefore, despite SRD5A2 inactivity, PEA may still support the recovery of neurosteroid levels through this alternative pathway. If there are any errors, feel free to leave another comment that we can further discuss.
Reference for my answer:
Locci, A. and Pinna, G. (2019) ‘Stimulation of PPAR-α by N-palmitoylethanolamine engages allopregnanolone biosynthesis to modulate emotional behavior’, Biological Psychiatry, available: https://www.researchgate.net/publication/331078884_Stimulation_of_PPAR-a_by_N-palmitoylethanolamine_engages_allopregnanolone_biosynthesis_to_modulate_emotional_behavior (you can download full pdf through this link)
This was a really interesting read, one point I would like to understand better is the extent to which PEA can meaningfully compensate for reduced 5-α-reductase activity. Since you mentioned that finasteride suppresses the enzyme involved in producing both DHT and allopregnanolone, I’m curious whether PEA primarily works by increasing synthesis indirectly through upstream precursors, or whether it can actually restore downstream neurosteroids even when the enzyme remains inhibited.